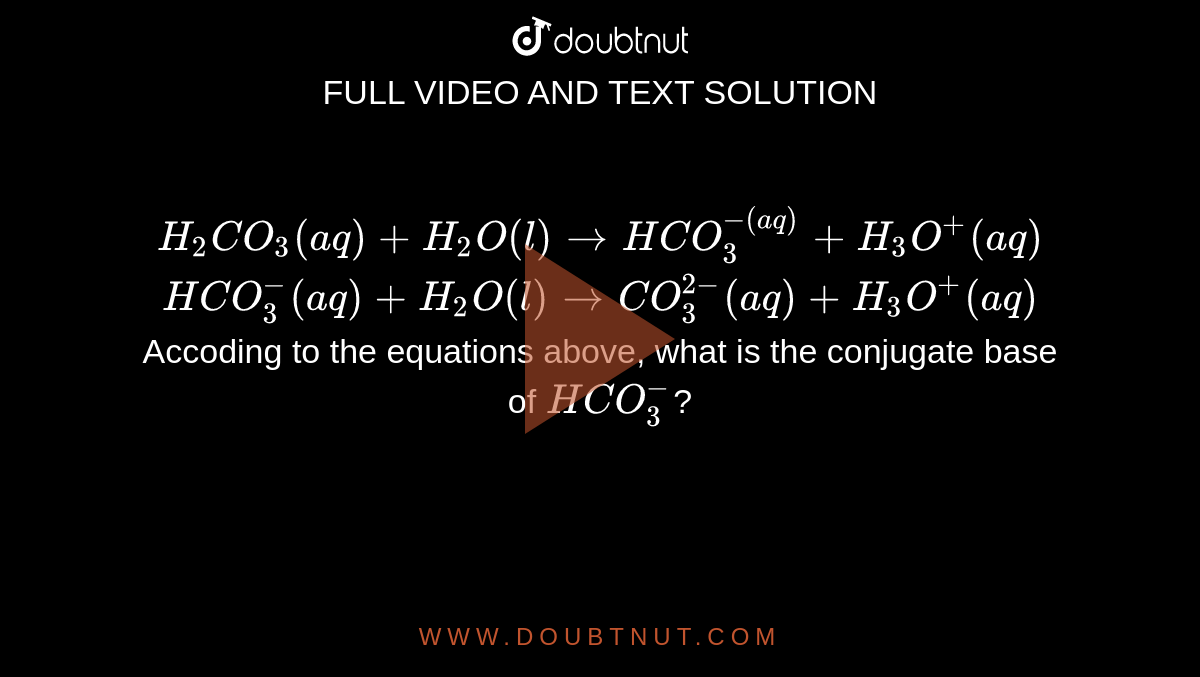
H2CO3(aq)+H2O(l)toHCO3^-(aq) + H3O^+(aq) HCO3^(-)(aq) + H2O(l)to CO3^(2-)(aq)+H3O^(+)(aq) Accoding to the equations above, what is the conjugate base of HCO3^-?

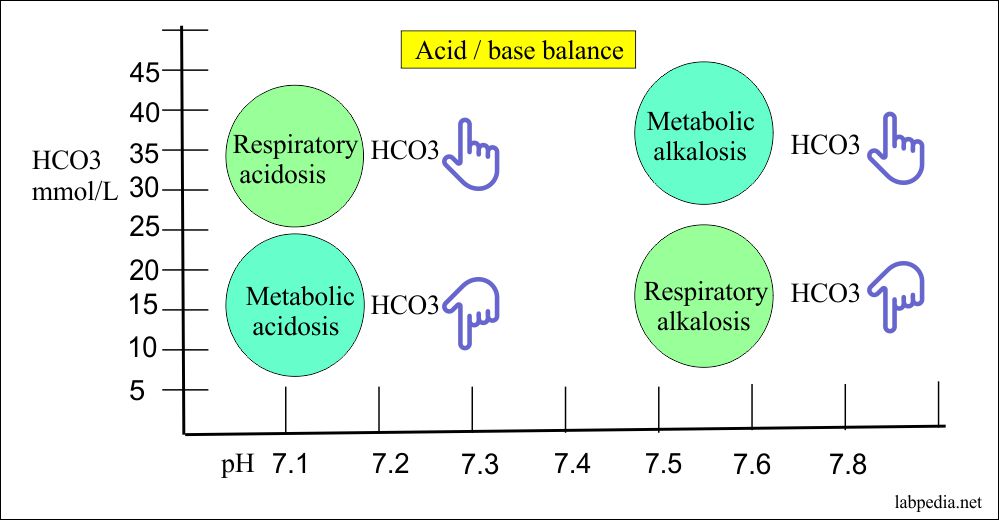
Rishi Kumar, MD - I created this table to teach my trainees how I approach acid-base problems assuming a normal bicarbonate (HCO3) of 24 mmol/L, PaCO2 40 mmHg, arterial pH 7.38-7.42, and



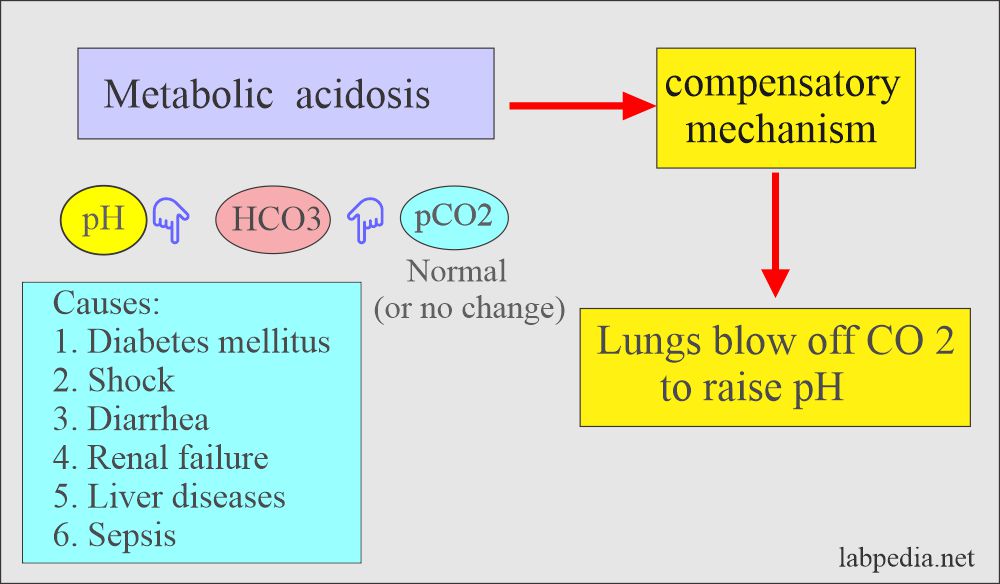
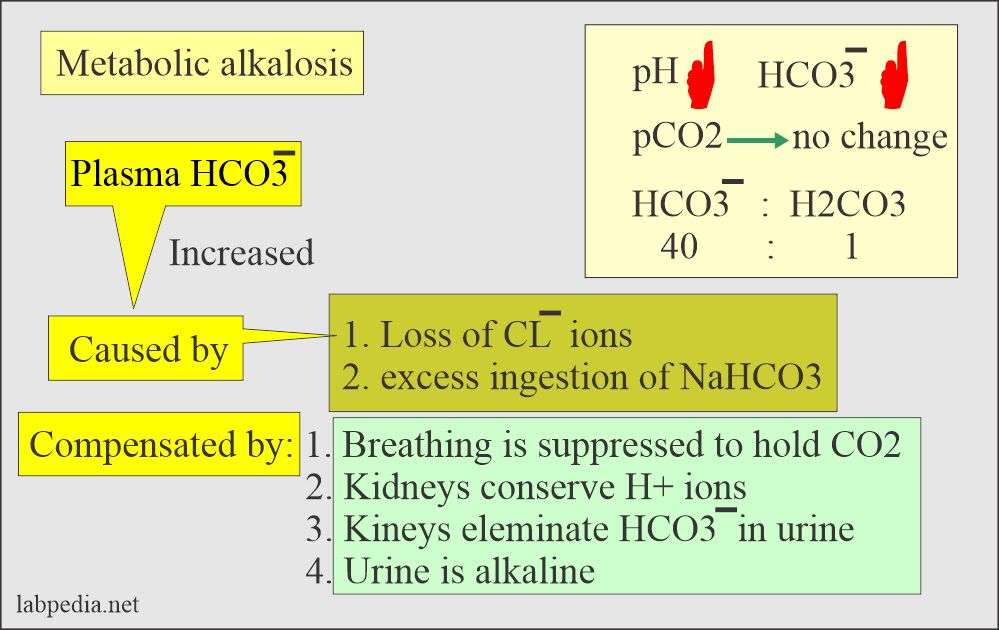



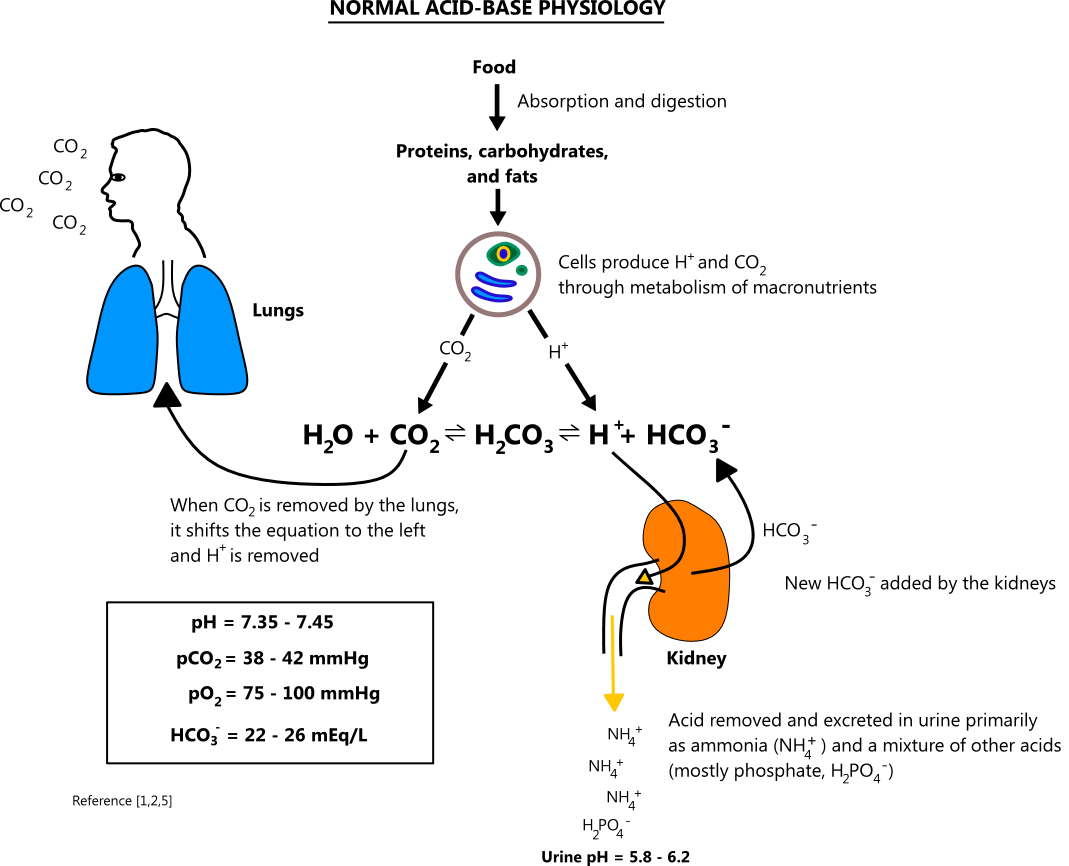
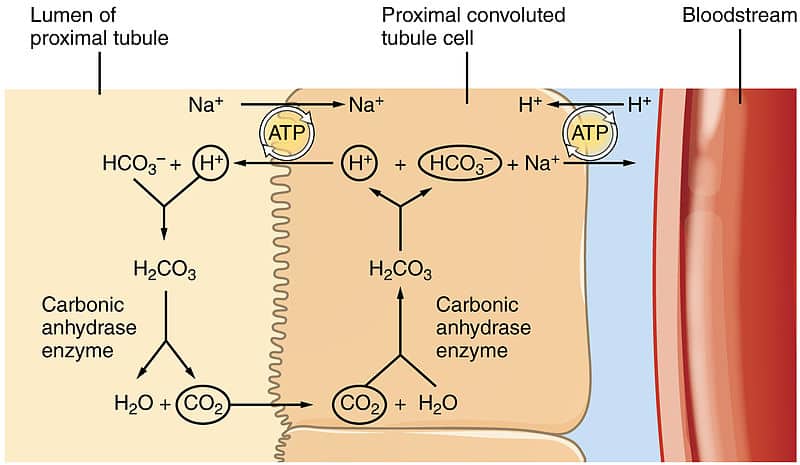

![ANSWERED] In the following reaction: HCO3(aq) + H₂... - Inorganic Chemistry ANSWERED] In the following reaction: HCO3(aq) + H₂... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/53160915-1659276540.7997923.jpeg)